The next big advance in fullerene chemistry: Opening up fullerene cages and inserting transition metals into them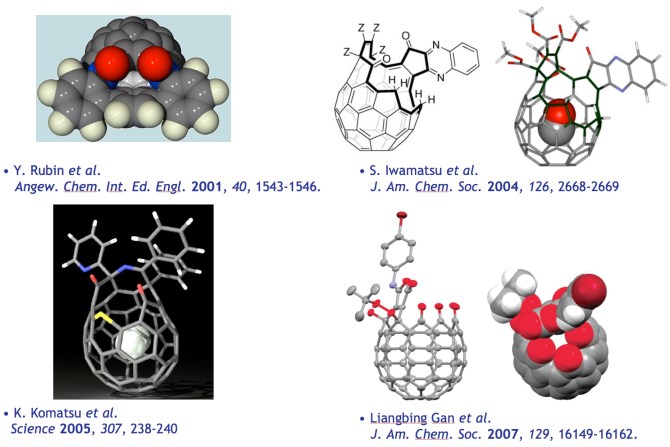
With their inner void of ≥3.5 Å, fullerenes have enough space inside them to accommodate any single atom of the periodic table (N, He, Ne, Ar, Kr, Xe, the lanthanide3+ cations)[1],[2] or a small molecule (H2, H2O, CO, NH3, Sc3N).[3],[4],[5],[6] Endohedral metallofullerenes encapsulating transition metals are the most exciting targets that have so far eluded three decades of attempted preparation. The various oxidation and spin states of these metals, coupled with the potentially strong electronic interaction with the carbon cage, make them ideal targets for synthesis. It is easily conceivable that these complexes will display a large array of electronic, magnetic, and photo-chemical properties that could be quite different from the currently known empty fullerenes or the lanthanide endohedral complexes (e.g. La@C82, Sc3N@C80). The potential for real-life applications for any of these compounds is very high.
We reported the first effectively open fullerene (1) and demonstrated that small gaseous atoms (He) or molecules (H2) can be incorporated under high pressure.[4] While we and others have developed synthetic methods to open fullerenes with larger orifices using complex reaction mechanisms, all known open fullerenes have resulted from unanticipated bond scission and fragmentation reactions. Currently, four known classes of open fullerenes have been discovered. None has an orifice that is wide enough for transition metal insertion.
We are currently pursuing an approach taking advantage of the [2+2+2] ring opening reaction of 1,2,3,4,5,6-hexaadducts of C60. This exciting approach has been challenging and has led to several highly encouraging results, which we are currently exploring.[7,8]
[1] a) “Stable Compounds of Helium and Neon: He@C60 and Ne@C60” Saunders, M.; Jiménez-Vázquez, H. A.; Cross, R. J.; Poreda, R. J. Science 1993, 259, 1428-1430. b) “Enrichment and Characterization of a Noble Gas Fullerene: Ar@C60” DiCamillo, B. A.; Hettich, R. L.; Guiochon, G., Compton, R. N.; Saunders, M.; Jiménez-Vázquez, H. A.; Khong, A.; Cross, R. J. J. Phys. Chem. 1996, 100, 9197-9201. c) “Observation of Atomlike Nitrogen in Nitrogen-Implanted Solid C60” Murphy, T. A.; Pawlik, T.; Weidinger, A.; Höhne, M.; Alcala, R.; Spaeth J.-M. Phys. Rev. Lett. 1996, 77, 1075-1078. d) “Isolation and Spectral Properties of Kr@C60, a Stable van der Waals Molecule” Yamamoto, K.; Saunders, M.; Khong, A.; Cross, R. J.; Grayson, M.; Gross, M. L.; Benedetto, A. F.; Weisman, R. B. J. Am. Chem. Soc. 1999, 121, 1591-1596. e) “Do Nitrogen-Atom-Containing Endohedral Fullerenes Undergo the Shrink-Wrap Mechanism?” Cao, B.; Peres, T.; Cross, R. J.; Saunders, M.; Lifshitz, C. J. Phys. Chem. A 2001, 105, 2142-2146. f) “129Xe NMR Spectrum of Xenon Inside C60” Syamala, M. S.; Cross, R. J.; Saunders, M. J. Am. Chem. Soc. 2002, 124, 6216-6219.
[2] a) “Endofullerenes: A New Family of Carbon Cluster”; Akasaka, T., Nagase, S., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2002. b) “Endohedral Clusterfullerenes—Playing with Cluster and Cage Sizes” Dunsch, L.; Yang, S. Phys. Chem. Chem. Phys. 2007, 9, 3067-3081.
[3] a) “Organic Approaches to Endohedral Metallofullerenes: Cracking Open or Zipping Up Carbon Shells?” Rubin, Y. Chem. Eur. J. 1997, 3, 1009-1016. b) “Ring Opening Reactions of Fullerenes: Designed Approaches to Endohedral Metal Complexes”; Rubin, Y. Top. Curr. Chem. 1999, 199, 67-91.
[4] “Insertion of Helium and Molecular Hydrogen Through the Orifice of an Open Fullerene”; Rubin, Y.; Jarrosson, T.; Wang, G.-W.; Bartberger, M. D.; Schick, G.; Saunders, M.; Cross, R. J.; Houk, K. N. Angew. Chem. Int. Ed. Engl. 2001, 40, 1543-1546.
[5] a) “100% Encapsulation of a Hydrogen Molecule into an Open-Cage Fullerene Derivative and Gas-Phase Generation of H2@C60” Y. Murata, M. Murata, and K. Komatsu, J. Am. Chem. Soc., 2003, 125, 7152-7153. b) “Synthesis, Structure, and Properties of Novel Open-Cage Fullerenes Having Heteroatom(s) on the Rim of the Orifice” Y. Murata, M. Murata, and K. Komatsu, Chem. Eur. J. 2003, 9, 1600-1609. c) “The Reaction of Fullerene C60 with 4,6-Dimethyl-1,2,3-Triazine: Formation of an Open-Cage Fullerene Derivative” Y. Murata, M. Murata, and K. Komatsu, J. Org. Chem. 2001, 66, 8187-8191. d) “Encapsulation of Molecular Hydrogen in Fullerene C60 by Organic Synthesis” K. Komatsu, M. Murata, Y. Murata, Science 2005, 307, 238-240.
[6] a) “A Bowl-Shaped Fullerene Encapsulates a Water into the Cage” Iwamatsu, S.-I.; Uozaki, T.; Kobayashi, K.; Re, S.; Nagase, S.; Murata, S.; J. Am. Chem. Soc. 2004, 126, 2668-2669. b) “Putting Ammonia into a Chemically Opened Fullerene”, Whitener Jr., K. E.; Frunzi, M.; Iwamatsu, S.; Murata, S. R.; Cross, J.; Saunders, M. J. Am. Chem. Soc. 2008, 130, 13996-13999.
[7] “Surgery of Fullerenes”, Murata, M.; Murata, Y.; Komatsu, K. Chem. Commun. 2008, 6083–6094.
[8] “Approaches to Open Fullerenes: A 1,2,3,4,5,6-Hexaadduct of C60”, Chuang, S.-C.; Clemente, F. R.; Khan, S. I.; Houk, K. N.; Rubin, Y. Org. Lett. 2006, 8, 4525-4528.
